Forward
This post represents yet another direction I want to push this blog…into the huge world of yeast and bacterial species you probably have never heard of. I think everyone that is serious about homebrewing eventually comes across a non-Saccharomyces species of yeast but 99% of the time, we are talking about Brettanomyces and now even entire breweries are devoted to it. This is the first of a series of posts will focus not only on Brettanomyces isolates and species but other yeasts you may not have heard of. I won’t just just talk about the properties of these yeasts but attempts to make a beer (or a fermented beverage) with them. I am still debating what form these beers will take but I think establishing a baseline for them is the best place to start. This series of posts will be yeast focused so the science will show up a little bit more than in the saison or straight brewing posts. As a bonus, you’ll probably get to hear me describe drinking some likely horrible tasting beers. This is a risk when you decide one day “I’m going to build a library of spoilage yeasts and ferment beers with pure cultures of them”…. That brings me to my final point, do not try this at home…OK, I won’t tell you what to do but whenever working with a microorganisms, learn as much about it as possible before proceeding. Some yeasts are pathogens and no one likes being sick. I am doing this so you don’t have to. (wink). I hope you enjoy the first of these installments…Kluyveromyces lactis.
Domain, Kingdom, Phylum, Class, Order, Family, Genus, Species, Sub species, Strain…see I am a biologist.
The most popular species used in beer production are members of two different genera, Saccharomyces and Brettanomyces. Within this blog, I’ve already begun to explore the Saccharomyces genus with the fermentation of Paradoxus ale, a beer that utilized Saccharomyces paradoxus (a close relative of S. cerevisiae). I also brewed some 100% Brettanomyces beers such as Brettanomyces clausseni Ale (although I may no longer believe that to be the case, this will be revisited in the near future). Let’s keep this microbiology thing going by pushing this a little further…
Both Saccharomyces and Brattanomyces are in the same family (and are closely related), Family Saccharomycetaceae. In addition to Saccharomyces and Brettanomyces, there are more than a dozen different genera in the family Saccharomycetaceae. All of these genera and species are closely related at the molecular level, although small differences in genetic makeup can result in wildly different phenotypes (physical characteristics). Species of the Candida genus can be pathogenic and are usually the source of yeast infections (specifically the species Candida albicans). People should be cautious straying too far outside of the Saccharomyces or Brettanomyces genera looking for interesting yeast to ferment with, but I do encourage experimentation.
Kluyveromyces is another genus in the family Saccharomycetaceae. Kluyveromyces lactis is an industrially important organism for its ability to ferment lactose into lactic acid. It is also used to make rennet (chymosin) for cheese production. My interest in this organism is the production of lactic acid (and the completed genome doesn’t hurt). Lactic acid is becoming more common in beer. Styles that are known for a strong lactic acid component are Berlinerweiss, Gose (pronounced gos-ay), Gueuze and other Lambic varieties…the sour stuff. The lactic acid is usually the product of Lactobacillus or Pediococcus species being present during the fermentation. Both Lactobacillus and Pediococcus are bacteria (prokaryotic), not yeast. These species are widely considered to be intolerant to compounds in hops, one of the reasons there aren’t very many hoppy sour beers. Weak acids found in hops such as trans-isohumulone, humulone, colupulone, and trans-humulinic acid inhibit Lactobacillus growth (primarily trans-isohumulone). Kluyveromyces lactis is already found in some fermented beverages. It was isolated from some burukutu back in the 1970s. Burukutu is a local beer in the Benue state of Nigeria (middle belt region near Cameroon), apparently rather popular due to how cheap it is. It is made with protein rich grains that grow easily in the region such as sorghum. It is alcoholic and sour, much like other spontaneously fermented beverages. I have never personally tried this beverage, although hope spring eternal, and I am not trying to replicate it…I’ll wait for Dogfish Head to do that for me.
What I do hope is that this can be a possible substitute for Lactobacillus in the fermentation of sour beers. There are several factors to consider. First, how does it grow? How sour can it make beer (pH)? Does it work in mixed fermentations? Is it tolerant to high levels of hops (isohumulone)? I won’t answer all these questions in this one post (that would make this a rather long and tedious post to read methinks), but I will do a few test batches with this organism over the course of the next several months and figure out what, if anything, can be done with this guy.
So what do we know about this organism? What flavor active compounds other than ethanol and lactic acid can it make? In previous posts, I’ve catalogued a variety of compounds made my Lactobacillus and Saccharomyces that may affect the final flavor of a beer. I did a literature dive to find out what, if any, compounds / secondary metaobolites Kluyveromyces lactis could make. This wasn’t easy and as far as I could tell, there has been no work done on analyzing K. lactis fermentation products when used in wort…just wine. So wine will have to do. There are obviously a different set of precursor molecules found in grape must than in wort…so the final product will be different. I tried to focus on the compounds that were likely going to be final products from wort as well…things derived from sucrose or simple sugars rather than plant specific compounds unlikely to be extracted from grains.
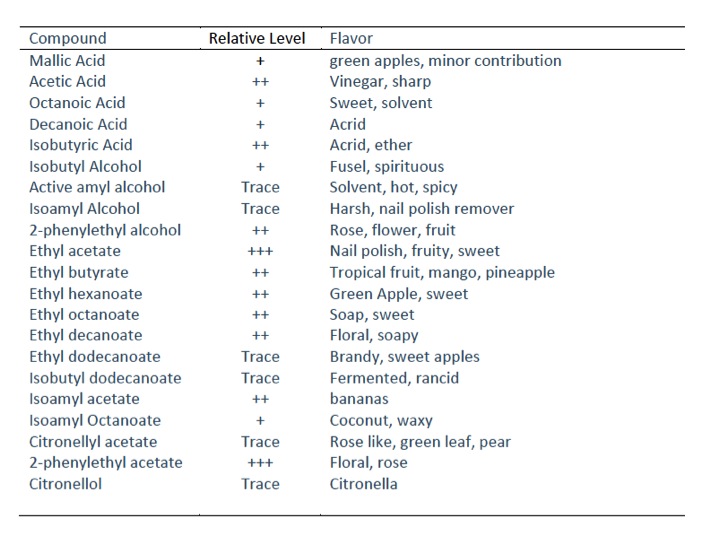
So the big surprises from my literature dive were acetic acid, pretty high concentrations, ethyl acetate and 2-phenylethyl acetate. This thing might not taste so bad after all…like a slightly sour saison yeast on steroids. Enough talk, onto the beer.
To start out, I’m going to be simple and brew 1 gallon of a simple wort, if it works out I’ll scale up and do some more experimentation with it. This will be a 100% Kluyveromyces lactis fermentation. Eventually, I want to do a mixed fermentation with some Dupont saison yeast and Kluyveromyces lactis, but I’m going to start out simple at first and let this series go wherever it wants to go. This yeast might be a killer strain, another reason to start slow.
Back story time: I’ve attempt a few different fermentations with this yeast and they all stalled out. I ended up over pitching it and it worked a little better. After months, I finally got a wort down to 1.010 FG. This is my starting point for this yeast. I tasted the batches that didn’t work out and I won’t comment on them here, I’m only going to focus on the first batch.
The Beer:
The wort of these experiments is the least interesting part, at least in these initial attempts. I decided to make a completely bland wort. It is 100% DME, OG 1.055 and IBU of 20 all from Saaz at 60 minutes.
Fermentation Conditions:
Fermentation took place in a 1 gallon glass jug (all of these will take place in there glass jugs between 65 and 80 F (it was sitting on the counter top in the kitchen for several months…no temperature control). Some interesting things happened during the fermentation. First, it formed a pellicle….of sorts.


The pellicle wasn’t like the dusty / bubble pellicles formed by some strains of Brettanomyces but this ropey, slimy mass that was physically attached to the wall of the jug. Even weirder, it went away and by the end it was basically gone. I’ve never seen a structure like this in a fermentation and been willing to let it ferment out all the way it can. A structure like this is usually a reason to worry, a reason to pour out the whole damn thing. I am pretty certain that this is a pure culture so I let it ride. This strain also fermented in cycles, it seemed to stop and then restarted. I had three separate krausen phases. I don’t think this was due to any contamination though. A repeat of this experiment will help confirm that suspicion.
I didn’t open the fermenter for the first several months, I wanted it to be as closed as possible to prevent contamination or at least minimize it. I use those three piece airlocks filled with water / sanitizer / alcohol so I can smell the off gasing easily as the fermentation is taking place. This particular fermentation consistently smelled like a basement or an old wet rug. When I was a kid, my mother was in medical school and we lived in this house in Missouri with a basement that leaked all the time. There was an old rug down in the basement and mushrooms would grow on it during the rainy season…it smelled like that. Mildew, dust, stale air, and metallic water. Not a pleasing aroma. I asked someone that worked with the yeast what it smelled like in the lab they said, “Like hell.” When I asked to elaborate on that with more descriptors she said, “Like socks you find in the bottom of a gym bag after a few weeks.” Old, wet, stale, gross. Pretty similar experience.
I originally received two different strains of this organism. For the purposes of my notes I am going to call them KL76 and KL92. Both of these strains have defined genotypes and all mutations have been mapped. I grew starters for both of these in 200 ml of YEPD media at 30° C for about 48 hours and then left the flasks sitting on a lab bench for several days. The cells settled out of solution. KL92 settled out of solution much faster compared to KL76. I took light absorbance readings at 600nm. KL76 gave an optical density of 0.131 and KL92 was 0.020. This is almost a 10 fold different in flocculation, I decided to move forward with the poorer floccing strain since I need it to be in solution. While choosing the one that flocced worse was a smart idea I feel, it was not sufficient. It still settled too quickly. On several occasions I would agitate the jug lightly to kick the bugs up into solution. They would fall out rather quickly, especially toward the end. I wanted it to get as low as it could so I kept agitating it to see if I could squeeze out a few more Plato from the wort.

The gravity settled out at 1.010, which is pretty good for an unknown organism. The hydrometer sample I took had lost that basement smell and actually had a pretty pleasant mildly acidic aroma to it. This may have turned out well after all.
Tasting Notes:
Well? Did I poison myself? Spoiler alert, I’m not dead.

I measured the pH using my meter. It was 3.8…so not sour. Beer is pretty clear, which isn’t too surprising since one of my problems with this yeast is its eagerness to fall out of solution, sorry for the personification. It is sort of amber, darker than I expected but not out of range by any means. This is uncarbonated, I haven’t bottled it and I won’t bother unless this tasting goes well. Then the bottles will be sent out to people who have already expressed interest in getting some of this.
Aroma: Aroma is pretty strong, it smells like sour apples…pink lady or granny smith. Has an almost chardonnay without the diacetyl sort of aroma, it is very fruity, a little acidic. No detectable acetic acid in the nose or lactic acid for that matter. I’m not sure if I should be surprised if this turns out not to have any lactic acid at all since I didn’t add lactose to the wort. There are mild hints of rubber…sulfur…in the nose, not off putting by any means but it is definitely there. Overall, it smells really nice. It smells like something I would drink honestly. It has some depth to it, the bright fruitiness is up front and in your face, it falls away pretty quickly to the earthy / rubber / sulfur. It’s sort of like having a good cheese on a plate of cut pears…if that makes any sense.
Beer is thin and a little flat on the flavor front. There is a mild aftertaste of grains, lingering apple flavors in the mouth. The rubber hits you first, falls away quickly to a white wine sort of feel to it. The aroma is far more complex than the taste…maybe that’s the wort’s fault. The second taste is better than the first, more fruit, more pears…some figs. Not sour…at all. The shocking thing about this beer is just the lack of body, you can barely tell you are tasting anything at all, then its gone.
I spoke with someone that tried to make a beer with this yeast years ago (by their own description they claimed they failed) and they said “It smells like a lot and tastes like nothing at all.” I would parrot that experience but not be so harsh. This beer begs for some element of sourness. If it were pH 3.2…this could be something special here. It has a lot of potential and I liked it…sort of.
Possible improvements and Upcoming Experiments:
Primarily I’d like to see how this yeast behaves in mixed fermentation compared to a Lactobacillus brevis maybe. If the final taste and mouthfeel of the beer could be improved upon, then you could have a really special beverage. Also, I’d like to test the hop tolerance of this strain compared to L. brevis or L. acidophilus. Mostly I just want to repeat this experiment and do it in parallel with a wort with some lactose in it. Let’s get this thing to sour up!
Upcoming brews:
Well, I’m committed to this experiment and series of posts now. Not sure how I’m going to deal with these posts moving forward but I already have Hanseniaspora uvarum, Torulaspora delbrueckii, Pichia membranifaciens, Metschnikowia pulcherrima, and Zygosaccharomyces bailii in hand but the next one will be a bacteria, Zymomonas mobilis. That one is going to be a hard post to write logistically but I’ll leave why that is to your imagination. The goal of this series of posts is to show the complexity, in isolation of one another ironically, of the pantheon of fermentative organisms. These organisms typically live in a niche someplace in the wort ecology…they don’t have to be the star of the fermentation to get noticed. By throwing them into the spotlight for a few fermentations, I may learn something new. Like the aroma of Kluyveromyces is incredible…taste contributions may be minimal.
“The mouth of a perfectly happy man is filled with beer.” — Ancient Egyptian Wisdom
Fantastic post, Matt, super interesting content!
Fantastic blog to read, congratulations! Keep experimenting and having fun. Best regards from Argentina.
Humberto Musa.
Almost makes me wish I was in DC, but I spent a summer living in College Park while working in Alexandria. It’s a nice place to visit (and drink), but I wouldn’t want to live there. 🙂
Very interesting post. I did my Ph.D in yeast genetics, albeit nothing to do with metabolism or fermentation, and always wondered if you could brew with K. lactis. You alluded to it, but I wonder if adding a little lactose to your starting wort, might bring some more complex flavors and sour it up a bit. Looking forward to n=2
Great experiment! I wonder if the bland flavor you lamented was due to the bland wort. If you used an all grain base with all Marris Otter, and something more interesting then Saaz to balance it, I wonder what you would have found. I get trying to simplify and avoid distractions from those other sources. But, I wonder how the yeast would have reacted to a denser grain, and if that would have supported the lacking mouth-feel you reported. I agree an experiment with stronger alpha acids is needed to evaluate this yeast, so that could really improve the beer (or prevent any fermentation). I look forward to reading more on your experience with this!
I’ve read that Kluyberomyces lactis cannot grow under anaerobic conditions. Sounds like the kinetics is limited by dissolved O2 so this maybe why it took so long to ferment to near-dryness. If this is the case, an aeration experiment would be fun. Great article…..best of luck!
I’ve read that Kluyveromyces lactis does not grow under anaerobic conditions. This may explain your long fermentation times. If this is the case, then an aeration experiment would be fun. Great article….Thanks!
Interesting experiment! Where did you hear that K. lactis produces lactic acid? It’s true that it can degrade lactose into glucose and galactose, but these enter the central carbon metabolism and are not converted into lactic acid. I guess that K. lactis can produce minor amounts of D-lactic acid via the same mechanism as S. cerevisiae, the methylglyoxal bypass, which is kind of a metabolic safeswitch. As a crabtree positive yeast, K. lactis does not produce ethanol in aerobic conditions and is unable to grow in fully anaerobic conditions. Here’s a good review if you are still interested in this organism: https://www.ncbi.nlm.nih.gov/pubmed/23576126
Thanks for the reply. I was operating under a few incorrect assumptions when I did this experiment. I appreciate the link.
Nevertheless, very interesting and valuable experiment. I was reading a blog post about an yeast species which indeed can form lactic acid and thought that you might be interested: http://beer.suregork.com/?p=3860
I received that yeast a few months back from the same source. I am working on a post of my own results. Might be up in a month or so.
Did you ever end up posting the follow up? I’ve been interested in experimenting with a sister strain (K. marxianus), and was interested in your results and sourcings for the strains.